What Did He Do to Ccr5 in Crispr Baby
- Research Highlight
- Open Access
- Published:
CCR5-Δ32 biology, gene editing, and warnings for the future of CRISPR-Cas9 as a human and humane gene editing tool
Cell & Bioscience volume x, Commodity number:48 (2020) Cite this commodity
Abstract
Background
Biomedical technologies take non but improved man wellness merely also assisted in the cosmos of human life. Since the commencement nascency of a good for you baby by in vitro fertilization (IVF) 40 years ago, IVF has been the mainstay treatment for couples struggling with infertility. This technology, in improver to increasingly accessible genetic testing, has made it possible for countless couples to have children. Since CRISPR-Cas9 gene editing was described in 2015, its potential for targeting genetic diseases has been much anticipated. However, the potential of using CRISPR-Cas9 for human germline modification has led to many fears of "designer babies" and widespread concerns for the impact of this applied science on human evolution and its implications in Social Darwinism. In add-on to these ethical/moral concerns, at that place remain many unknowns nigh CRISPR-Cas9 technology and endless unanticipated consequence to gene editing.
Methods
In this paper, we clarify the current progresses of CRISPR-Cas9 technology and talk over the theoretical advantages of certain allelic variances in the C-C chemokine receptor five gene (CCR5) in the setting of recent upstanding/moral concerns regarding gene editing using the CRISPR-Cas9 organisation.
Results
These uncertainties take been highlighted recently by the birth of Chinese twins whose C-C chemokine receptor 5 (CCR5) cistron had been inactivated via CRISPR-Cas9 to exist theoretically protective against HIV infection. CCR5 signaling is critical for the successful infection of man immunodeficiency virus (HIV) and people with homozygous inactivating CCR5-Δ32 mutations have been shown to be protected against HIV infection. Those with the CCR5-Δ32/Δ32 mutation also accept greater neuroplasticity, assuasive for improved recovery from neurological trauma, and decreased Chagas cardiomyopathy. However, the CCR5-Δ32/Δ32 mutation has besides been associated with earlier clinical manifestations for West Nile infection, ambiguous effects on osteoclast role, and a four-fold increased mortality from flu infection. These detrimental health impacts, in addition to the confounding factor that these CRISPR babies do not behave this exact CCR5-Δ32/Δ32 mutation, lead to many questions regarding the children's time to come health and the moral puzzler of their nativity. The creation and nascence of these babies was not completed with any scientific, ethical, or governmental oversight, which has spurned the dispatch of talks regarding global regulations for homo genetic editing.
Conclusions
Although we can try to regulate for ethical, health-related only apply of this technology, moral and governmental oversights need to be supplemented by technical regulations. For instance, whole genome sequencing needs to exist used to eliminate off-target mutations that could affect the health and safety of infants born to this process. Like Pandora's Box, we cannot pretend to forget CRISPR-Cas9 applied science, all we can exercise is ensure a safe, moral, and equitable used of this technology.
Every bit the nigh efficient and precise genome editing tool bachelor, CRISPR-Cas9 technology presents a powerful and lost-price method of genetic editing that has never been bachelor earlier. The availability of this technique has radically changed the biomedical field and has the potential to radically change homo healthcare [1,2,three]. It has fabricated in vitro modeling of human being mutations possible, increased the speed of genetically engineered beast models, and made the handling of genetic diseases more a pipedream. In fact, a airplane pilot clinical trial in sickle jail cell anemia just reported promising preliminary results in the first patient ever treated with CRISPR-Cas9 gene-therapy [4] and there are multiple other ongoing trials assessing factor therapy in hematologic disease [5]. The ability of CRISPR-Cas9 technology is not limited to the correction of disease-causing genetic mutations, but also being considered as a method for taking advantage of genetic traits inherent in some populations. For instance, the C-C chemokine receptor 5 (CCR5) Δ32 mutation found in ~ xi% of northern Europeans is known to protect against HIV infection. Last year, twin Chinese girls were engineered past CRISPR-Cas9 to conduct a CCR5 gene with like backdrop to CCR5-Δ32, specifically to be resistant to HIV. The announcement of these unexpected births has highlighted the fear of a new era of eugenics brought on by CRISPR-Cas9. Here we discuss the protective and detrimental effects of this mutation and contribute to the ongoing moral, philosophical, and regulational conversation with considerations regarding the technical safe of CRISPR-Cas9 technique in humans.
The CCR5 factor was first identified in 1977 [6] simply did not get a subject of bully public interest until 2009, when an HIV positive individual transplanted with bone marrow from a donor with a homozygous CCR5-Δ32 mutation, became HIV negative despite stopping anti-retroviral (ARV) therapy [7]. This seminal clinical case report was founded on decades of work showing CCR5'southward role as a co-stimulator in T-prison cell function, activation, and the production of antigen specific T-cells [8]. These studies showed the CCR5-Δ32 mutation to crusade deletion of 32-base pairs in CCR5, leading to non-functional expression of this gene that does not localize to the prison cell surface. These mechanistic findings along with the discovery of CCR5 as a necessary co-receptor for entry of macrophage tropic HIV strains [9, 10] led to increased interest in this gene as a target for HIV treatment and other immunological processes.
CCR5 deletions have also been shown to provide protection against other pathogens, including smallpox and flaviviruses such as dengue, Zika, and West Nile virus [11]. In fact, smallpox endemics in Europe are believed to be the selective force per unit area that led to an increased presence of the allele in European populations [11]. CCR5 deletion was also found to be protective against not-viral infections. Early reports have plant the CCR5 -Δ32 deletion to exist protective confronting inflammatory cardiomyopathy in patients with chronic Chagas' illness [12]. This result was recently disputed in a polymorphism assay between wild-type, heterozygous, and homozygous Chaga'southward illness patients [13]. However, a Brazilian genetic polymorphism report of CCR1, CCR5, and their ligands CCL2 and CCL5, respectively, constitute CCL5-CCR1 to be the target for allowed-stimulation from Trypanosoma cruzi infection. Certain variants of CCL5-CCR1 were subsequently found to be significantly protective against Chagas'due south disease [14]. Exterior of the infectious disease realm, CCR5 has also been found to exist involved in neuronal recovery from stroke and traumatic brain injury (TBI) through upregulation of CREB (cAMP response chemical element-binding poly peptide) and DLK (Delta-similar poly peptide 1) signaling [15]. Joy et al. first identified the expression of CCR5 in cortical neurons after stroke and later discovered neuronal knockdown of CCR5 to result in enhanced cortical projections during regeneration and preservation of dendritic spines [15]. These in vitro findings were subsequently confirmed as clinically significant in an assay of 1,563 stroke patients (300 CCR5-Δ32 carriers vs 1265 non-carriers) in the Tel Aviv Brain Acute Stroke Cohort (TABASCO). Patients with Δ32/Δ32 loss-of-function mutation CCR5 recovered significantly faster from stroke with improved measures of memory, exact office, and attending- indicating improved neuronal plasticity [15]. While CCR5 is clinically relevant in this wide variety of diseases, its importance in HIV infection has been the most studied in the clinical setting.
As a cell membrane integrated protein with seven transmembrane segments and an eighth α-helix parallel to the plasma membrane, CCR5 presents on the cell surface and functions in tandem with CD4-recptors as the initial co-docking site for the HIV PG120-PG41 surface protein. This initial association betwixt the HIV PG120-PG41, CCR5, and CD4-receptors allows for the initial viral invasion and subsequent infection and replication (Fig. 1a). The essential binding site on CCR5 for HIV PG120-PG41 is known as 2D7. It is located on the third extracellular element (second loop) of the membrane integrated CCR5 and works in tandem with the PA12 binding site and the 1000 protein linkage domains found on the first extra-cellular element of CCR5. The CCR5-Δ32 mutation, describes a 32 base pair deletion just before the 2D7 structural loop. This results in the creation of a premature stop codon, and thus, the absence of the 2D7 loop necessary for HIV viral bounden, simply preserves the PA12 bounden site (Fig. 2). This mutation hampers HIV binding two-fold: by removing the necessary 2D7 binding domain and past rendering the protein cytosolic. Around 10% of the European population take paired missense mutations C20S and C178R or C101X and FS299, collectively known equally CCR5-Δ32, which protects against HIV infection by inhibiting the initial viral docking procedure (Fig. 1b) [16, 17].
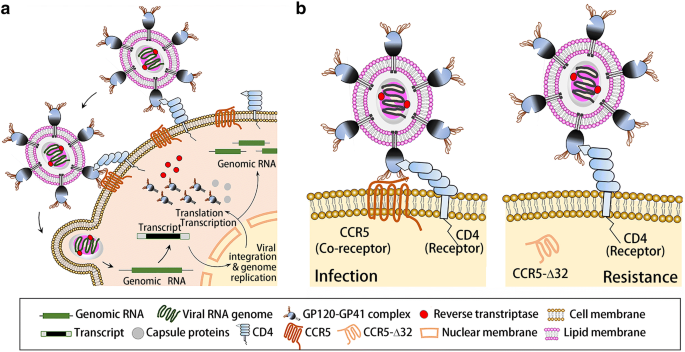
The HIV infection process (a): The HIV GP-120 first associates with both the CD4 and CCR5 on the surface of a cell, which is the showtime step in viral invasion and further viral replication. Molecular mechanism of CCR5 in HIV infection and the protective effect of cytoplasmic CCR5-Δ32 against HIV-1 infection (b)
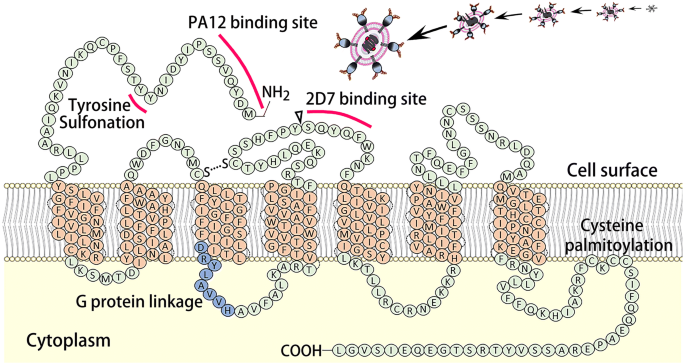
The construction of membrane integrated CCR5. The elements important in HIV binding and structure (PA12 binding site and 2D7 binding site, and sites of tyrosine sulfonation and G-protein linkage) are highlighted. The CCR5-Δ32 deletion site is denoted with a triangle and plant merely earlier the 2D7 binding site. Mutation at this site results in a premature finish codon, and thus the deletion of all protein structures after this location, resulting in the loss of the 2D7 binding site and a cytosolic CCR5
Ever since the theoretical protection of CCR5-Δ32/Δ32 against HIV was clinically supported by the cure of a HIV-positive patient transplanted with os marrow from a homozygous CCR5-Δ32 donor [vii], the potential for CCR5-Δ32 every bit a curative therapy for HIV has been greatly debated and anticipated [8, 17, eighteen]. However, most controlled and regulated studies are still in the pre-clinical phase using human stalk cells or mouse models. The Deng group established a CRISPR/Cas9 gene editing system in human CD34+ hematopoietic stalk cells (HSPCs) which immune for long-term CCR5 ablation. Mice transplanted with these CCR5-deleted HSPCs exhibited lasting HIV-1 resistance in vivo [19]. Some other study plant editing of co-receptors CCR5 and CXCR4 by CRISPR-Cas9 to protect CD4+ T cells from HIV-1 infection in vitro [xx]. Although some other group was able to successfully transplant and achieve long-term engraftment of CRISPR-edited HSPCs into a patient, they were simply able to disrupt 5% of CCR5 role. This unexpected result hinted at unanticipated factors in in vivo editing, thus halting the report for fear of damage to patient health [21, 22]. Despite the lack of complete understanding of the CCR5 gene and incomplete pre-clinical testing provingCCR5 cistron manipulation to be benign, some have already jumped alee to human genome manipulation. Last yr, Jiankui He, a researcher at the Southern Academy of Science and Technology in Guandong, Prc announced the birth of twins whose genomes he had manipulated by CRISPR-Cas9 to have non-functional CCR5. This editing was made in an effort to protect the infants confronting HIV infection. This unregulated experiment immediately generated massive business organisation over the moral affect of this human being experiment and earned universal condemnation for advancing to man experimentation without adequate condom precautions and assessments.
While the use of CRISPR-Cas9 engineering science equally a eugenics tool is morally confounding and hard to justify given the human health, evolution, and social equality implications; it is naïve to say that CRISPR-Cas9 will not be used past futures parents and scientist to give an advantageous foundation to their children. Thus, the all-time course of activeness that global summits on genome editing can produce are exact allowances and restrictions for genome editing and specific punishments for both the researcher and the local/federal governments responsible for enforcing regulations. Inherited disease caused by specific betoken mutations may be the most realistic targets for germline alternation. For instance, correcting the point mutation causing the glutamine to valine mutation in sickle cell disease could complimentary future generation from the constant threat of pain crises and eliminate the risk of acute chest and stroke that often claim these patients' lives. However, even in these clear-cut cases we still demand further information on the exact fourth dimension menstruation during which germline alteration is safe for the embryo. Yet, to ensure at least the methodological safety of using CRISPR-Cas9 in humans, two technical aspects must be met: full agreement of the gene being altered and consummate command over off target furnishings of CRISPR-Cas9 editing. Editing of CCR5 does non fit the first requirement as those homozygous for the CCR5-Δ32 mutation take unexpected negative furnishings such as earlier clinical manifestations for West Nile infection [23], 4-fold likelihood of mortality from influenza infections [24], and disadvantageous osteoclast function [25]. In addition, multiple publications have reported unexpected off-target mutations generated by CRISPR-Cas9. Although ane retracted publication demonstrated few unexpected mutational events [26], one study found rare simply notable mutations [27], several others constitute big deletions [28, 29], while another found unexplainable complex deletions and insertions in mice generated past CRISPR-Cas9 [30]. As such, the CCR5 twins need to exist monitored both for possible known effects, such as an increased susceptibility to influenza infection, abnormal bone growth and other immunological conditions, and also require close monitoring of their general growth and development for unanticipated effects.
Even should these unknowns be overcome, there may all the same be pocket-size deletions or insertions that crusade deleterious frame-shift mutations, or rarer effects we have all the same to identify. Equally such, the only manner to ensure the coding fidelity of edited cells is by sequencing the full genome of each edited cell in comparison to parents' genomes. This safety check itself volition crave further technological evolution allowing for rapid, cheap whole-genome sequencing and analysis while in the narrow window of implantable embryos. Even these precautions would not account for the epigenetic factors that may touch growth and development. Should complications from these identified elements be resolved, there are all the same a myriad of unknown factors in CRISPR-Cas9 technology that should nowadays an independent technological precaution confronting homo genetic editing regardless of the moral/upstanding conundrum (Fig. iii). We suggest that there needs to exist a more vigorous and annual global debate to established the specific mutations on which man gene-editing enquiry should be allowed, and that these genes be limited to those what would solve articulate clinical issues (i.e. sickle cell disease, other diseases with known mutational causes). Ideally, such a body of experts would too be able to propose a multinational consortium such as the Un on the appropriate punitive and incentive deportment necessary to dissuade individuals and institutions from supporting unsanctioned human genome editing.

Advantages and disadvantages of CCR5-Δ32/Δ32 (a) and CRISPR-Cas9 (b) genome editing
Availability of data and materials
Non applicable.
Abbreviations
- ARV:
-
Anti-retroviral
- CCR5:
-
C-C chemokine receptor 5
- CCL2:
-
C-C motif chemokine ligand ii
- CREB:
-
cAMP response element-binding protein
- CRISPR:
-
Clustered regularly interspaced curt palindromic repeats
- Cas9:
-
CRISPR-associated protein-9 nuclease
- CXCR4:
-
C-X-C chemokine receptor type iv
- DLK1:
-
Delta-similar protein one
- GP120:
-
Glycoprotein 120, viral envelope glycoprotein 120
- GP41:
-
Glycoprotein 41, viral envelope glycoprotein 41
- HSPC:
-
Hematopoietic stem prison cell
- HIV:
-
Human immunodeficiency virus
- IVF:
-
In vitro fertilization
- TABASCO:
-
Tel Aviv Brain Acute Stroke Cohort
- TBI:
-
Traumatic brain injury
References
-
Republic of mali P, Yang L, Esvelt KM, Aach J, Guell M, DiCarlo JE, Norville JE, Church GM. RNA-guided man genome engineering via Cas9. Science. 2013;339(6121):823–6. https://doi.org/10.1126/scientific discipline.1232033.
-
Cong 50, Ran FA, Cox D, Lin S, Barretto R, Habib N, Hsu PD, Wu X, Jiang W, Marraffini LA, Zhang F. Multiplex genome engineering using CRISPR/Cas systems. Science. 2013;339(6121):819–23. https://doi.org/10.1126/science.1231143.
-
Fonfara I, Richter H, Bratovič Thou, Le Rhun A, Charpentier E. The CRISPR-associated DNA-cleaving enzyme Cpf1 too processes precursor CRISPR RNA. Nature. 2016;532(7600):517–21. https://doi.org/10.1038/nature17945.
-
Stein R. Gene-edited 'Supercells' make progress In fight Against Sickle Cell Disease. National Public Radio. https://www.npr.org/sections/health-shots/2019/11/19/780510277/gene-edited-supercells-brand-progress-in-fight-against-sickle-prison cell-affliction. Accessed 19 Nov 2019.
-
Demirci S, Uchida Northward, Tisdale JF. Cistron therapy for sickle cell disease: an update. Cytotherapy. 2018;xx(7):899–910. https://doi.org/10.1016/j.jcyt.2018.04.003.
-
Walz DA, Wu VY, de Lamo R, Dene H, McCoy LE. Primary construction of human platelet cistron 4. Thromb Res. 1977;11:893–8. https://doi.org/10.1016/0049-3848(77)90117-7.
-
Hütter M, Nowak D, Mossner M, Ganepola S, Müßig A, Allers K. Long-term control of HIV by CCR5 Delta32/Delta32 stem-cell transplantation. North Engl J Med. 2009;360:692–8. https://doi.org/ten.1056/NEJMoa0802905.
-
Barmania F, Pepper MS. C-C chemokine receptor blazon five (CCR5): An emerging target for the control of HIV infection. Appl Transl Genom. 2013;2:iii–16. https://doi.org/ten.1016/j.atg.2013.05.004.
-
Dragic T, Litwin V, Allaway GP, Martin SR, Huang Y, Nagashima KA. HIV-1 entry into CD4+ cells is mediated by the chemokine receptor CC-CKR-5. Nature. 1996;381:667–73. https://doi.org/ten.1038/381667a0.
-
Deng H, Liu R, Ellmeier Due west, Choe S, Unutmaz D, Burkhart Yard. Identification of a major co-receptor for primary isolates of HIV-i. Nature. 1996;381:661–half dozen. https://doi.org/x.1038/381661a0.
-
Galvani A, Slatkin M. Evaluating plague and smallpox as historical selective pressures for the CCR5-Δ32 HIV-resistance allele. PNAS. 2003;100(25):15276–9. https://doi.org/10.1073/pnas.2435085100.
-
Flórez O, Martín J, González CI. Genetic variants in the chemokines and chemokine receptors in Chagas disease. Hum Immunol. 2012;73(8):852–8. https://doi.org/10.1016/j.humimm.2012.04.
-
Oliveira AP, Bernardo CR, Camargo AV, Villafanha DF, Cavasini CE, de Mattos CC, de Godoy MF, Bestetti RB, de Mattos LC. CCR5 chemokine receptor gene variants in chronic Chagas' disease. Int J Cardiol. 2014;176(2):520–2. https://doi.org/10.1016/j.ijcard.2014.07.043.
-
Batista AM, Alvarado-Arnez LE, Alves SM, Melo G, Pereira IR, Ruivo LAS, da Silva AA, Gibaldi D, da Silva TDESP, de Lorena VMB, de Melo AS, de Araújo Soares AK, Barros MDS, Costa VMA, Cardoso CC, Pacheco AG, Carrazzone C, Oliveira Westward Jr, Moraes MO, Lannes-Vieira J. Genetic polymorphism at CCL5 is associated with protection in Chagas' heart disease: antagonistic participation of CCR1+ and CCR5+ cells in chronic Chagasic cardiomyopathy. Front end Immunol. 2018;9:615. https://doi.org/x.3389/fimmu.2018.00615.
-
Joy MT, Ben Assayag E, Shabashov-Rock D, Liraz-Zaltsman S, Mazzitelli J, Arenas M, Abduljawad N, Kliper E, Korczyn AD, Thareja NS, Kesner EL, Zhou Chiliad, Huang South, Silva TK, Katz Due north, Bornstein NM, Silva AJ, Shohami E, Carmichael ST. CCR5 is a therapeutic target for recovery afterward stroke and traumatic brain injury. Cell. 2019;176(v):1143–57. https://doi.org/10.1016/j.prison cell.2019.01.044.
-
Parmentier 1000. CCR5 and HIV infection, a view from Brussels. Front Immunol. 2015;6:295. https://doi.org/10.3389/fimmu.2015.00295.
-
Allers K, Schneider T. CCR5Δ32 mutation and HIV infection: basis for curative HIV therapy. Curr Opin Virol. 2015;14:24–9. https://doi.org/ten.1016/j.coviro.2015.06.007.
-
Hütter G, Bodor J, Ledger S, Boyd M, Millington M, Tsie Grand, Symonds G. CCR5 targeted cell therapy for HIV and prevention of viral escape. Viruses. 2015;7(8):4186–203. https://doi.org/10.3390/v7082816.
-
Xu 50, Yang H, Gao Y, Chen Z, Xie L, Liu Y, Liu Y, Wang Ten, Li H, Lai West, He Y, Yao A, Ma Fifty, Shao Y, Zhang B, Wang C, Chen H, Deng H. CRISPR/Cas9-mediated CCR5 ablation in homo hematopoietic stem/progenitor cells confers HIV-1 resistance in vivo. Mol Ther. 2017;25(viii):1782–ix. https://doi.org/ten.1016/j.ymthe.2017.04.027.
-
Liu Z, Chen S, Jin X, Wang Q, Yang K, Li C, Xiao Q, Hou P, Liu S, Wu Due south, Hou W, Xiong Y, Kong C, Zhao X, Wu L, Li C, Sun 1000, Guo D. Genome editing of the HIV co-receptors CCR5 and CXCR4 by CRISPR-Cas9 protects CD4+ T cells from HIV-i infection. Jail cell Biosci. 2017;seven:47. https://doi.org/10.1186/s13578-017-0174-2.
-
Xu 50, Wang J, Liu Y, Xie L, Su B, Mou D, Wang L, Liu T, Wang X, Zhang B, Zhao Fifty, Hu L, Ning H, Zhang Y, Deng Thou, Liu Fifty, Lu X, Zhang T, Xu J, Li C, Wu H, Deng H, Chen H. CRISPR-edited stem cells in a patient with HIV and astute lymphocytic leukemia. N Engl J Med. 2019;381(thirteen):1240–vii. https://doi.org/x.1056/NEJMoa1817426.
-
Wei X, Nielsen R. CCR5-Δ32 is deleterious in the homozygous country in humans. Nat Med. 2019;25(half-dozen):909–10. https://doi.org/10.1038/s41591-019-0459-6.
-
Lim JK, McDermott DH, Lisco A, Foster GA, Krysztof D, Follmann D, Stramer SL, Irish potato PM. CCR5 deficiency is a run a risk factor for early clinical manifestations of West Nile virus infection but non for viral transmission. J Infect Dis. 2010;201(ii):178–85. https://doi.org/10.1086/649426.
-
Falcon A, Cuevas MT, Rodriguez-Frandsen A, Reyes Northward, Pozo F, Moreno S, Ledesma J, Martínez-Alarcón J, Nieto A, Casas I. CCR5 deficiency predisposes to fatal outcome in flu virus infection. J Gen Virol. 2015;96:2074–eight. https://doi.org/10.1099/vir.0.000165.
-
Xie Y, Zhan S, Ge Westward, Tang P. The potential risks of C-C chemokine receptor 5-edited babies in os development. Os Res. 2019;vii:ane–iv. https://doi.org/10.1038/s41413-019-0044-0.
-
Schaefer KA, Wu WH, Colgan DF, Tsang SH, Bassuk AG, Mahajan VB. Unexpected mutations after CRISPR–Cas9 editing in vivo. Nat Methods. 2017;xiv(half-dozen):547–8. https://doi.org/10.1038/nmeth.4293(Retracted).
-
Iyer V, Shen B, Zhang W, Hodgkins A, Keane T, Huang X, Skarnes WC. Astray mutations are rare in Cas9-modified mice. Nat Methods. 2015;12(six):479. https://doi.org/10.1038/nmeth.34088.
-
Adikusuma F, Piltz Southward, Corbett MA, Turvey M, McColl SR, Helbig KJ, Beard MR, Hughes J, Pomerantz RT, Thomas PQ. Large deletions induced by Cas9 cleavage. Nature. 2018;560:E8–E9. https://doi.org/10.1038/s41586-018-0380-z.
-
Ma H, Marti-Gutierrez North, Park SW, Wu J, Lee Y, Suzuki M, Koski A, Ji D, Hayama T, Ahmed R, Darby H, Van Dyken C, Li Y, Kang E, Park AR, Kim D, Kim ST, Gong J, Gu Y, Xu X, Battaglia D, Krieg SA, Lee DM, Wu DH, Wolf DP, Heitner SB, Belmonte JCI, Amato P, Kim JS, Kaul S, Mitalipov South. Correction of a pathogenic gene mutation in human embryos. Nature. 2017;548:413–9. https://doi.org/10.1038/nature23305.
-
Shin HY, Wang C, Lee HK, Yoo KH, Zeng X, Kuhns T, Yang CM, Mohr T, Liu C, Hennighausen 50. CRISPR/Cas9 targeting events cause complex deletions and insertions at 17 sites in the mouse genome. Nat Commun. 2017;31(eight):15464. https://doi.org/10.1038/ncomms15464.
Funding
There is no funding back up for this study. The drove of references for this article is supported by the Medical Scientist Training Programme (MSPT), NIH.
Author information
Affiliations
Contributions
MX conceived and prepared the manuscript. The author read and approved the terminal manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed in the discussed studies.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional data
Publisher'due south Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Admission This article is licensed under a Creative Eatables Attribution 4.0 International License, which permits use, sharing, accommodation, distribution and reproduction in any medium or format, as long equally you requite appropriate credit to the original author(s) and the source, provide a link to the Artistic Eatables licence, and indicate if changes were made. The images or other third political party material in this article are included in the article's Creative Eatables licence, unless indicated otherwise in a credit line to the material. If textile is not included in the article'south Creative Commons licence and your intended employ is not permitted by statutory regulation or exceeds the permitted utilize, you volition need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/iv.0/. The Artistic Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/nothing/i.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the information.
Reprints and Permissions
Almost this article
Cite this article
Xu, Thousand. CCR5-Δ32 biological science, gene editing, and warnings for the future of CRISPR-Cas9 equally a man and humane gene editing tool. Prison cell Biosci 10, 48 (2020). https://doi.org/10.1186/s13578-020-00410-vi
-
Received:
-
Accepted:
-
Published:
-
DOI : https://doi.org/10.1186/s13578-020-00410-6
Keywords
- CCR5 -Δ32
- Man genome editing
- HIV infection
- CRISPR-Cas
Source: https://cellandbioscience.biomedcentral.com/articles/10.1186/s13578-020-00410-6
Post a Comment for "What Did He Do to Ccr5 in Crispr Baby"